CMC Development
Category: *CRO
Exhibitor: SHANGHAI MEDICILON INC.
Booth No: M1124
Characteristic
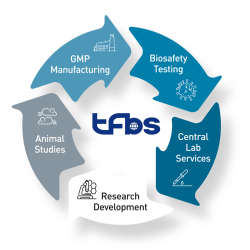
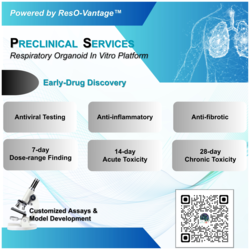
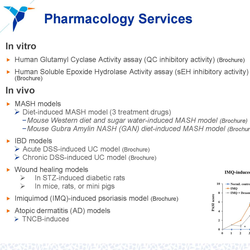
Medicilon provides integrated Chemistry, Manufacturing, and Controls (CMC) services supporting drug development from preclinical research through IND submission, clinical trials, and early commercialization. By combining API process development, drug product formulation, analytical & quality systems, and GMP manufacturing, we deliver a true one-stop CMC platform designed to reduce regulatory risk, accelerate timelines, and preserve long-term development flexibility.
Other Products
Products you may be interested in
Highest Rated Products