Translational Medicine
Category: *CRO
Exhibitor: QPS HOLDINGS, LLC
Booth No: M1213
Characteristic
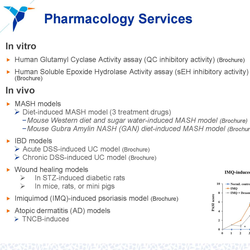
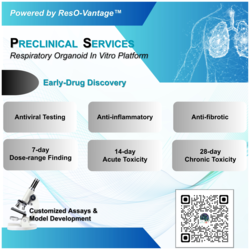
Quantitation projects in translational medicine include qualification and validation of commercially available kits and methods; method transfer of assays originated from the Sponsor’s lab; independent method development; or a collaborative effort between the QPS team and your lab for custom assays. In addition to immunoassays for PK, biomarkers, and immunogenicity assessment, QPS can develop cell-based assays for neutralizing antibody (NAb) activity studies and biomarker evaluation in support of your drug development programs.
Other Products
Products you may be interested in
Highest Rated Products