TSY-310 - A Next-Generation Novel Bispecific ADC
Model: TSY-310
Category: Pharmaceutical
Exhibitor: FORMOSA PHARMACEUTICALS , INC
Booth No: M318
Characteristic
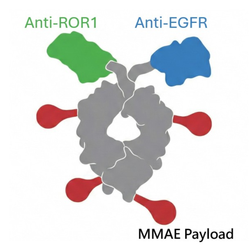
√ Innovative Bispecific ADC: A bispecific Fc-fusion antibody-drug conjugate (Bispecific ADC) featuring a dual-targeting design specifically for EGFR and ROR1, which can selectively kill tumor cells while reducing damage to normal cells.
√ Targeting Solid Tumors such as NSCLC: Its primary therapeutic indication is for solid tumors like Non-Small Cell Lung Cancer (NSCLC), which accounts for up to 85% of all lung cancer cases.
√ Breakthrough Molecular Structure: Featuring a "single-chain architecture," its size is only about half that of a traditional monoclonal antibody, providing deeper tumor penetration capabilities, simplifying the manufacturing process, and prolonging its half-life.
√ Overcoming Targeted Therapy Resistance: When cancer cells develop resistance to EGFR therapies, their ROR1 expression often increases abnormally; TSY-310 can proactively identify and attack these resistant cells, filling the gap of unmet medical needs left by current therapies.
√ High Safety Profile and Bystander Effect: Since ROR1 is barely expressed in normal adult tissues, TSY-310 is endowed with high specificity and safety; furthermore, paired with the clinically validated MMAE toxin, it has demonstrated significant anti-tumor activity and a "bystander effect" in preclinical trials.
√ Capturing the Blue Ocean Market of Bispecific ADCs: Currently, there are no approved "bispecific ADCs" on the global market; amidst the boom of ADC licensing deals, TSY-310 possesses high scarcity, innovative potential, and promising commercial value.
Other Products
Products you may be interested in
Highest Rated Products