Drug Product
Category: *CDMO
Exhibitor: CARBOGEN AMCIS
Booth No: L524
Characteristic
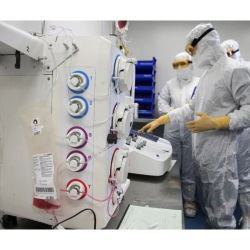
The development and manufacturing of sterile drug products require skills and long-term experience in pharmaceutical sciences, microbiology, engineering and process development.
This expertise extends from drug formulation and sterilization methods to aseptic pharmaceutical manufacturing. A solid understanding of regulatory standards is also requested for safe development and production of sterile drug products following Good Manufacturing Practices (GMP) and other applicable guidelines.
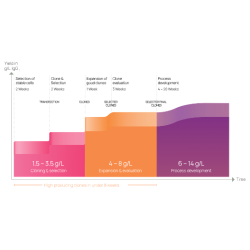
Drug product CDMO with more than 20 years of expertise
Specialized in the development and manufacturing of sterile drug products
Offering both liquid and lyophilized drug products
Brand new for clinical and commercial supply, Annex 1 & GMP-compliant facility compliant
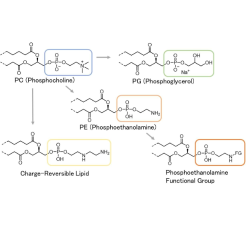
Handling complex compounds, formulations and high-potency compounds
Other Products
Products you may be interested in
Highest Rated Products